The Science Behind Pulse Wave™ Focused Shockwave Therapy
A deep dive into the physics, engineering, and clinical science that power Pulse Wave focused electromagnetic shockwave therapy — built for providers who want to understand what separates this technology from everything else on the market.
The Science Behind Pulse Wave Therapy | Focused Shockwave Physics for Providers
What Is Focused Shockwave Therapy?
A plain-language introduction for providers new to focused shockwave technology — what it is, who uses it, and why it matters clinically.
The Simple Version
Focused Shockwave Therapy (FSWT) is a non-invasive treatment that uses precisely aimed acoustic pressure waves to deliver mechanical energy deep into injured tissue — without surgery, injections, or downtime.
Think of it as applying targeted mechanical stress to a specific point inside the body. That stress triggers your patient's own healing biology: new blood vessel growth, collagen remodeling, inflammation reset, and nerve regeneration.
Who Uses It
Focused shockwave therapy is used by physical therapists, chiropractors, orthopedic surgeons, sports medicine physicians, and regenerative medicine specialists. It has a strong evidence base for musculoskeletal conditions including plantar fasciitis, tendinopathies, calcific tendinitis, and non-union fractures.
Focused vs. Radial — A Key Distinction
Radial (pressure wave) devices scatter energy broadly across tissue. Focused shockwave converges energy at a precise point at a specific depth — like a magnifying glass vs. a light bulb. This precision is what enables treatment of deep structures and produces the strongest clinical evidence.
Conditions Treated
- OrthopedicPlantar fasciitis, calcific tendinitis, Achilles, patellar, rotator cuff tendinopathies
- Sports MedicineHamstring insertional, lateral epicondylitis (tennis elbow), stress fractures
- RegenerativeChronic non-healing wounds, avascular necrosis, non-union fractures
- UrologyPeyronie's disease, erectile dysfunction (vascular etiology)
- NeurologySpasticity reduction, neurogenic pain modulation
FDA Registration
The Pulse Wave device is FDA Class I registered and manufactured in Denver, CO under ISO 13485 quality management systems. Providers can use it with confidence in a clinical setting.
Shockwave Anatomy & Wave Dynamics
Every Pulse Wave focused shockwave therapy pulse follows a precise pressure cycle. Understanding this waveform is the foundation for evaluating any device's therapeutic potential.
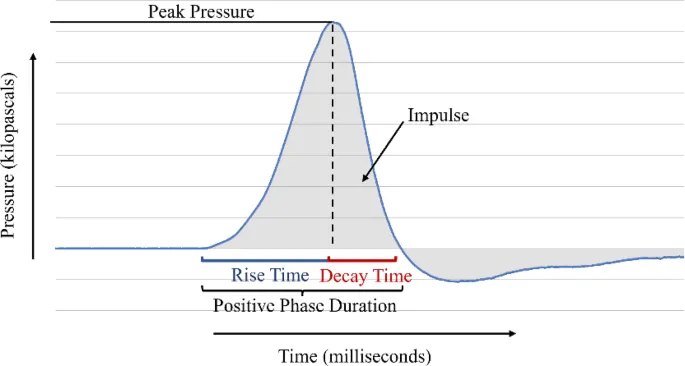
Rise time is how fast the pulse climbs from ambient pressure to peak. Measured in nanoseconds, a steeper rise time indicates a sharper, more focused energy front — a hallmark of true focused shockwave technology.
True shockwaves have sub-microsecond rise times. This is what distinguishes them from radial pressure waves, which have much slower rise times and cannot focus energy at depth.
After reaching peak pressure, the Pulse Wave focused shockwave rapidly falls back toward zero. This positive phase is the primary therapeutic driver, delivering compressive mechanical force into tissue to trigger biological repair cascades including mechanotransduction and growth factor release.
The wave dips below baseline into negative pressure territory before returning to rest. This tensile phase contributes to cavitation — the formation and collapse of microbubbles in fluid-rich tissue — which plays a key role in cellular stimulation, membrane permeability changes, and tissue repair.
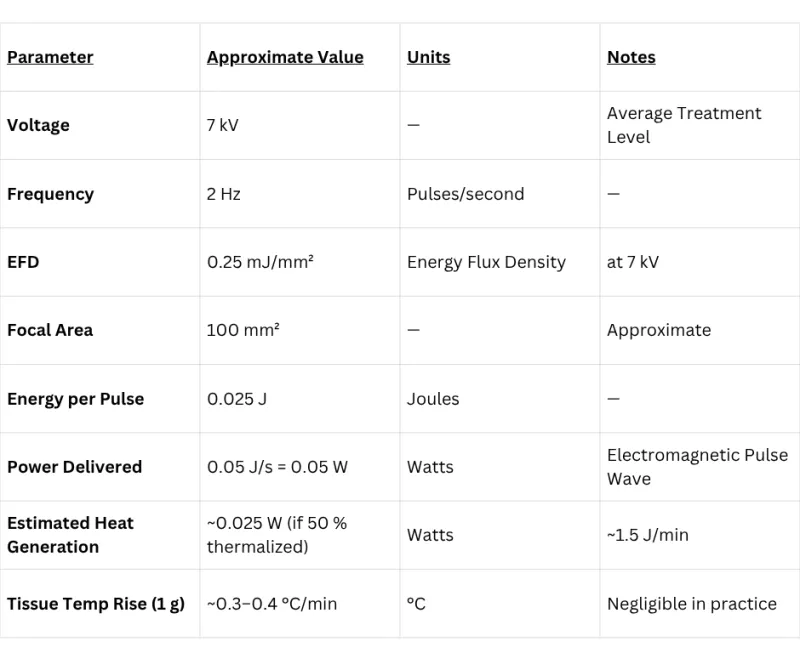
Focused shockwave therapy power is measured in kilovolts (kV), where 1 kV = 1,000 volts. Higher voltage settings on the Pulse Wave device generate stronger peak pressures, giving providers precise control over treatment intensity for different clinical applications from pain modulation to deep tissue stimulation.
Pulse Wave Technology — Patented Stepped Parabolic Reflector
The engineering innovations that make Pulse Wave focused shockwave therapy different from every other device on the market.
Patent-Pending Stepped Parabolic Reflector
Pulse Wave's proprietary stepped parabolic geometry focuses the electromagnetic waveform from the point of generation — eliminating the acoustic lens found in cylindrical and flat-coil EM devices. Near 100% energy reaches the focal target.
Dual-Waveform Output
The stepped parabolic design simultaneously generates two therapeutic waveforms — an electromagnetic pulse (EMP) and a true focused shockwave — from a single applicator head. No second device needed.
Made in Denver, CO
Every Pulse Wave device is manufactured in Denver, Colorado under ISO 13485 quality management. FDA Class I registered. No overseas electronics, no long repair turnarounds.
No Consumable Electrodes
Unlike electrohydraulic systems that require electrode replacement, Pulse Wave uses only distilled water for maintenance. 1,000,000+ impulse applicator lifespan with no degradation in waveform quality.
What the Stepped Parabolic Reflector Means Clinically
Conventional EM devices use a separate acoustic focusing lens that absorbs a portion of the energy before it reaches the patient. Pulse Wave's pre-focused parabolic geometry eliminates this loss — meaning every joule of energy generated is delivered to the therapeutic target, not lost in the focusing element.
How Pulse Wave Focused Shockwave Therapy Works
From electromagnetic pulse generation to acoustic wave delivery — a step-by-step breakdown of the Pulse Wave mechanism.
Step 1: Electromagnetic Generation
A high-voltage capacitor discharges through a flat coil behind a metallic membrane. The resulting electromagnetic pulse repels the membrane outward, generating a planar acoustic wave — the raw energy source for every Pulse Wave therapy pulse.
Step 2: Parabolic Reflector Focusing
The planar wave strikes the stepped parabolic reflector surface. Unlike a standard parabolic reflector, the stepped design pre-focuses the wave from the moment of generation — the geometric shape itself creates the focal convergence, with no acoustic lens required.
Step 3: TrueDepth™ Adjustable Depth
The Pulse Wave device's proprietary TrueDepth system allows providers to adjust the focal depth from the skin surface to 60mm deep within tissue — without changing applicator heads. Turn a dial, shift the focal point. Every clinical indication is covered with a single applicator.


Why Focal Depth Flexibility Matters
A device locked at a fixed focal depth forces the provider to choose between surface and deep treatments. Pulse Wave's 60mm TrueDepth range means you can treat superficial structures (plantar fascia, epicondyle insertions) and deep structures (hip joint, lumbar facets, deep rotator cuff) with the same applicator in the same session.
Depth & Treatment Zones by Condition
Matching focal depth to the target structure is critical for effective focused shockwave therapy. Pulse Wave's TrueDepth system covers every clinical scenario.

Superficial (10–20 mm)
Plantar fascia, Achilles insertion, lateral epicondyle, patellar tendon insertion, superficial calcifications. Low-to-medium EFD settings sufficient.
Mid-Depth (20–40 mm)
Rotator cuff (supraspinatus), greater trochanteric bursitis, proximal hamstring insertion, tibialis posterior. Medium-to-high EFD for adequate energy at depth.
Deep (40–60 mm)
Hip joint capsule, lumbar facet joints, deep hip flexors, sacroiliac ligaments. Pulse Wave's 60mm TrueDepth is required — competing devices often cannot reach these targets.
Focused Shockwave Device Types — A Complete Comparison
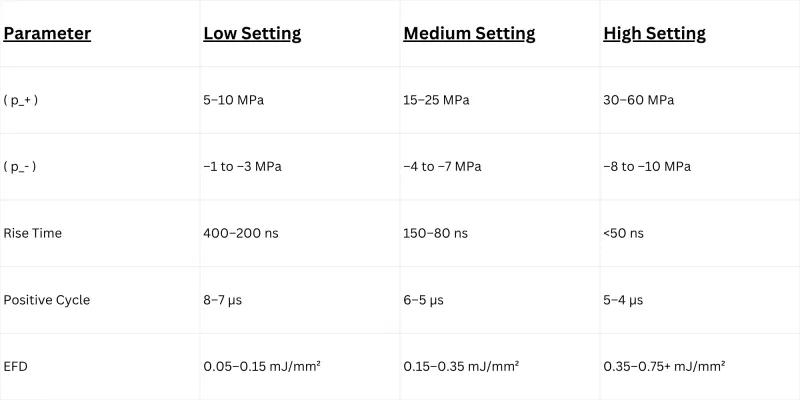
Three generator technologies dominate the focused shockwave market. Only one is still evolving. Here's what providers need to know before buying.
The oldest focused shockwave technology, using spark-gap discharge developed in the 1950s–70s. Works by discharging a high-voltage spark across electrodes submerged in a saline-filled reflector bowl.
The Problems:
- Saline corrodes electrodes — waveform consistency degrades with every use
- Approximately 50% of energy is lost before reaching the target due to inefficient generation and saline coupling losses
- Electrodes are consumables requiring regular replacement at ongoing cost
- Saline water management (mixing, refilling, disposal) adds clinical overhead
- Spark-gap technology is inherently inconsistent — each spark is slightly different
Why Competitors Still Sell It:
Electrohydraulic devices have a long clinical history and existing research base. Some manufacturers buy the technology from overseas suppliers for $10,000–$20,000 and sell units in the US for $55,000–$80,000+. The markup funds marketing, not engineering.
⚠ Buyer Awareness
If a vendor is selling you a focused shockwave device with electrodes and saline in 2025, ask them what the annual electrode replacement cost is, and request an independent EFD measurement at treatment 1 vs. treatment 500. The waveform degradation data is not flattering.
Uses hundreds to thousands of piezoelectric ceramic crystals arranged on a spherical surface. Each crystal deforms slightly when charged, and the combined acoustic output converges at the focal point.
Advantages: Very precise focal point, minimal maintenance, quiet operation, consistent waveform.
Limitations: Applicator heads are fragile and expensive to replace if damaged. Generally limited to low-to-medium energy ranges. Large applicator head can limit access to anatomical sites.
Piezoelectric devices are well-suited for musculoskeletal pain applications but may not reach the peak pressures needed for calcific tendinitis fragmentation or deep structure treatment.
Electromagnetic generators use a coil and membrane system to generate acoustic waves. Three distinct reflector designs exist — and the differences matter significantly:
Cylindrical Reflector: High power, but energy must be refocused by a separate acoustic lens, introducing energy loss at the lens interface.
Flat Coil: Generates an unfocused planar wave that requires a separate focal lens. Two-step focusing means additional energy scatter.
Stepped Parabolic (Pulse Wave): The newest and most efficient design. The reflector geometry itself creates focused output from generation — no acoustic lens, no two-step focusing, near 100% energy delivery to the target.
Pulse Wave: Patent-Pending Stepped Parabolic Reflector
Generates two simultaneous therapeutic waveforms (electromagnetic pulse + true focused shockwave) from a single applicator. No consumables, no saline, no electrode replacement — just distilled water maintenance. 1,000,000+ impulse rated applicator life with consistent waveform from first to last pulse.
Head-to-Head Device Comparison
| Feature | Electrohydraulic | Piezoelectric | EM Parabolic (Pulse Wave) |
|---|---|---|---|
| Technology Era | 1950s–70s spark-gap | 1980s ceramic array | Latest generation |
| Consumables | Electrodes + saline (ongoing cost) | None (fragile heads) | None (distilled water only) |
| Waveform Consistency | Degrades with electrode wear | Excellent | Excellent, 1M+ impulses |
| Energy Efficiency | ~50% lost (saline + lens) | Good | ~100% to focal target |
| Maintenance Overhead | High (electrode replacement, saline) | Low (costly if cracked) | Minimal (distilled water) |
| Dual Waveform | No | No | Yes — EMP + FSW simultaneously |
| Max Focal Depth | Varies (typically 40–60mm) | Typically 30–50mm | 60mm TrueDepth adjustable |
| US Price Range | $55,000–$80,000+ | $40,000–$70,000+ | Contact for provider pricing |
| Pulse Wave Device | — | — | ✓ Stepped Parabolic Reflector |
📈 The Price Markup Reality
Many competitors purchase electrohydraulic or generic EM devices from overseas manufacturers at $10,000–$20,000 and sell them in the US at $55,000–$80,000. You're paying for the brand and the sales team, not the engineering. Pulse Wave is manufactured domestically in Denver, CO — the margin goes to R&D and US manufacturing quality, not overseas arbitrage.
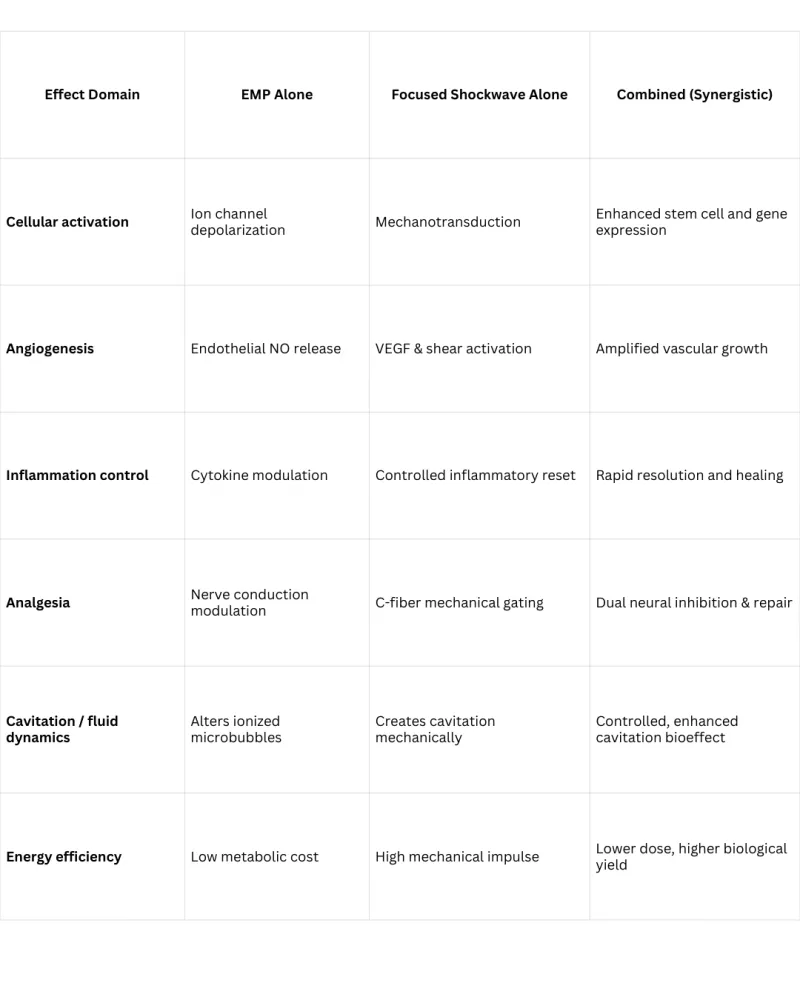
Dual-Wave Therapy — Two Mechanisms, One Applicator
Pulse Wave's patented stepped parabolic reflector generates two distinct therapeutic waveforms simultaneously from a single applicator — delivering more therapeutic pathways per treatment session.
Electromagnetic Pulse (EMP)
- Cellular membrane polarization — primes cells for repair
- Enhanced microcirculation — improves local blood flow
- Inflammatory signaling modulation
- Neuromodulation and pain reduction
- Mast cell degranulation (anti-inflammatory)
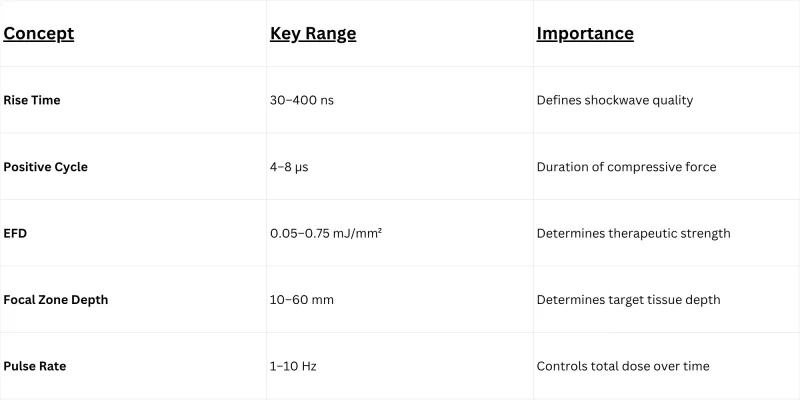
- Rise Time30–400 ns
- Magnetic Field0.1–2 Tesla (localized)
True Focused Shockwave
- Mechanotransduction and gene expression activation
- Angiogenesis and neovascularization
- Inflammatory reset and tissue remodeling
- Nerve regeneration and analgesia
- Calcification fragmentation
- EFD Range0.008–0.33 mJ/mm²
- Peak PressureUp to 38 MPa
- TrueDepth60mm adjustable focal depth
The Clinical Implication
With Pulse Wave therapy, every pulse delivers two complementary biophysical mechanisms simultaneously — without adding treatment time, complexity, or a second device. Providers who previously used separate PEMF and shockwave devices can consolidate to one applicator with enhanced synergistic effects.
How Pulse Wave's Dual Waveforms Work Together
Three proposed coupling effects explain why delivering both waveforms simultaneously may produce enhanced clinical outcomes compared to sequential mono-therapy.
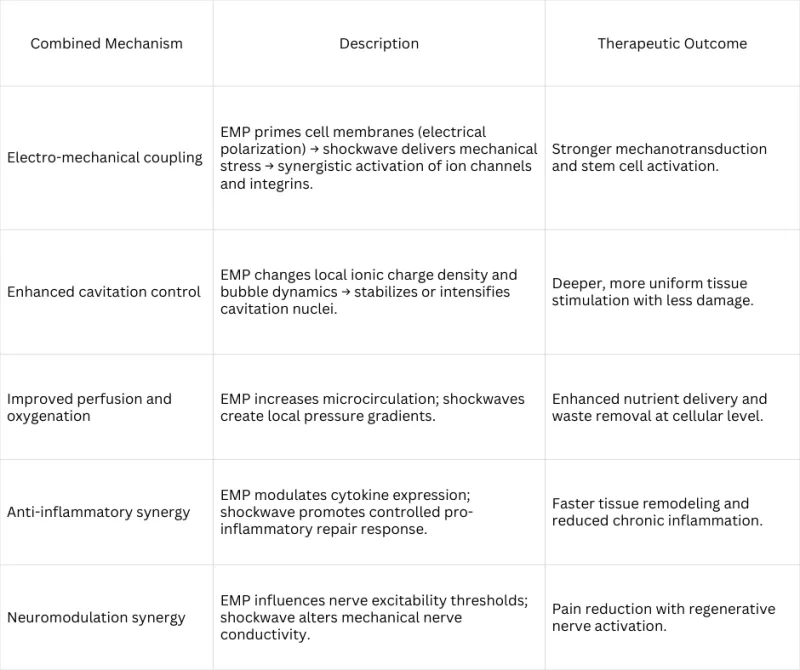
Sequential Activation Model
The electromagnetic pulse arrives first and preconditions cellular membranes — increasing membrane permeability and priming ion channel activity. When the focused shockwave arrives microseconds later, cells are in a heightened responsive state, potentially amplifying mechanotransduction signaling.
Field Coupling Effect
The electromagnetic and acoustic wavefronts propagate through the same tissue volume simultaneously. Where these fields overlap at the focal zone, they may constructively interact — the EM field altering the mechanical behavior of the tissue in ways that enhance acoustic wave absorption and biological uptake.
Enhanced Cavitation
The electromagnetic field alters bubble nucleation thresholds during the shockwave's negative pressure phase, potentially lowering the threshold for therapeutic cavitation and amplifying the micro-mechanical stimulation that drives cellular repair responses.
What This Means for Your Practice
With Pulse Wave therapy, you deliver two complementary mechanisms in every pulse — without adding treatment time, complexity, or a second device. One applicator, one session, two pathways to healing. Providers treating chronic non-responders to radial shockwave often see meaningful outcomes with Pulse Wave's dual-waveform approach.
Complete Technical Specifications
Full specification table for the Pulse Wave focused electromagnetic shockwave therapy system — for clinicians, biomedical engineers, and procurement teams.
| Parameter | Specification |
|---|---|
| Physical / Device | |
| Manufacturer | Pulse Wave Medical |
| Manufacturing Location | Denver, Colorado, USA |
| Regulatory Status | FDA Class I Registered |
| Quality Management | ISO 13485 Certified |
| Applicator Life | 1,000,000+ impulses |
| Power & Electrical | |
| Input Power | 100–240 VAC, 50/60 Hz |
| Power Consumption | <500 W peak |
| Generator Type | Electromagnetic (flat coil) |
| Voltage Range | Adjustable (low → high kV) |
| Acoustic Wave Parameters | |
| Wave Type | True focused shockwave (acoustic) |
| Peak Pressure | Up to 38 MPa at focal point |
| Energy Flux Density (EFD) | 0.008 – 0.33 mJ/mm² |
| Frequency Range | 1 – 5 Hz (adjustable) |
| Rise Time | <1 µs (true shockwave) |
| Focusing System | |
| Reflector Design | Patent-pending stepped parabolic reflector |
| Focusing Mechanism | Geometric pre-focus (no acoustic lens) |
| Energy Efficiency | Near 100% to focal target |
| Focal Depth (TrueDepth™) | 0 – 60 mm (continuously adjustable) |
| Focal Zone Dimensions | Ellipsoid; depth-dependent |
| Electromagnetic Pulse (EMP) | |
| EMP Rise Time | 30 – 400 ns |
| Magnetic Field Intensity | 0.1 – 2 Tesla (localized, focal zone) |
| EMP Waveform | Simultaneous with shockwave (single pulse) |
| Applicator & Coupling | |
| Coupling Medium | Distilled water (internal, no saline required) |
| Consumables | None (no electrodes, no saline mixing) |
| Applicator Configuration | Single applicator, dual-output |
| Contact Interface | Ultrasound gel (standard clinical) |
| Certifications & Compliance | |
| FDA Registration | Class I, 510(k) exempt |
| ISO Certification | ISO 13485:2016 |
| EMC Compliance | IEC 60601-1 |
| Electrical Safety | IEC 60601-1 |
Request Full Spec Sheet
For complete technical documentation, IFU (Instructions for Use), or biomedical engineering specifications, contact Shockwave Therapy Pro at tyler@shockwavetherapypro.com or call (801) 520-5177.
Provider Support
Have questions about Pulse Wave focused shockwave therapy physics, device specifications, clinical applications, or pricing? We're the official US distributor.

